
Lewis Dot Diagram Of Hcn
See the diagram below: Now you can see that the central atom here is Carbon because it is easy for Carbon to become stable as it is the least electronegative of all. However, hydrogen is the least electronegative but it cant be a central atom because it has only one spare electron.
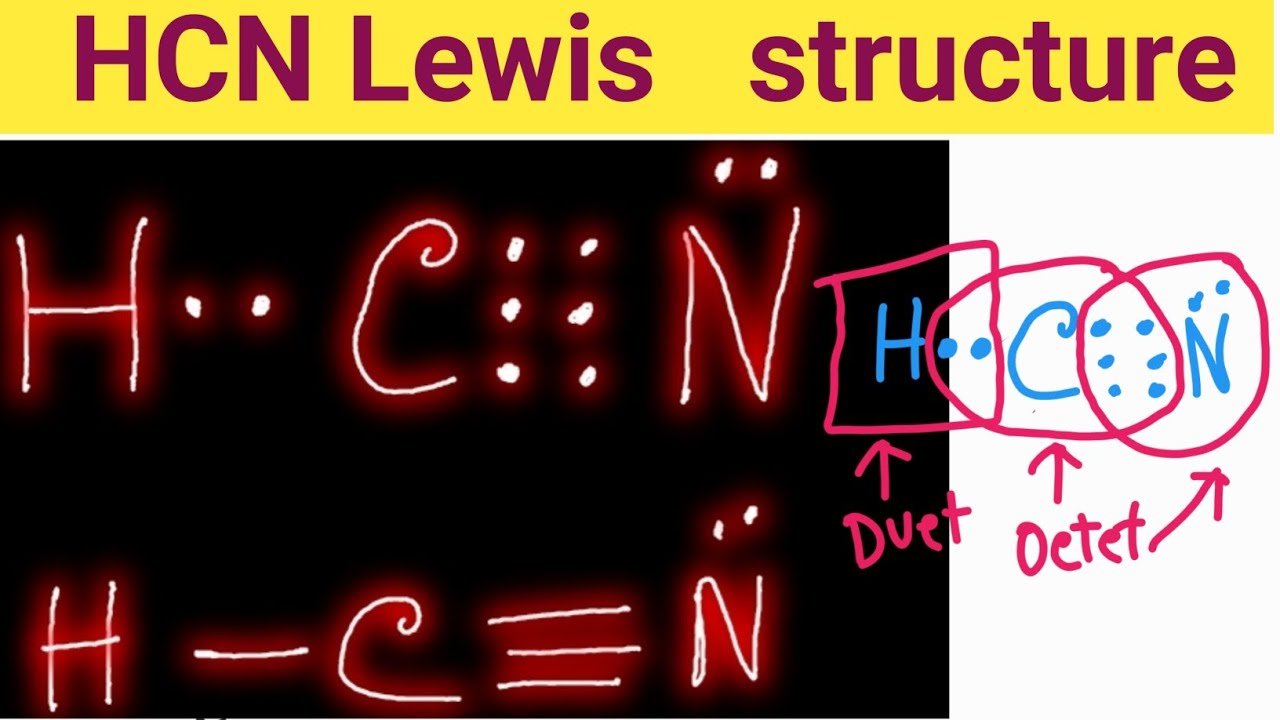
HCN Lewis StructureHydrogen Cyanide (HCN) Lewis Dot StructureDraw Lewis Structure of HCN
Drawing Lewis diagrams. A Lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. When constructing a Lewis diagram, keep in mind the octet rule, which refers to the tendency of atoms.
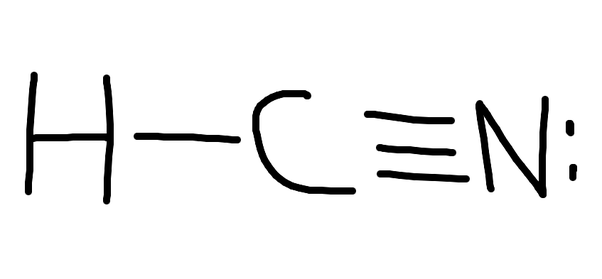
Lewis Diagram For Hcn
What is this molecule and what is it used for? HCN, hydrogen cyanide, is rather poisonous. HCN is a gas used primarily in chemical synthesis, mining, and polymer manufacturing. But serious, it's dangerous, so stay away unless you are a legit chemist. Method 1: Step method to draw the Lewis structure of HCN.
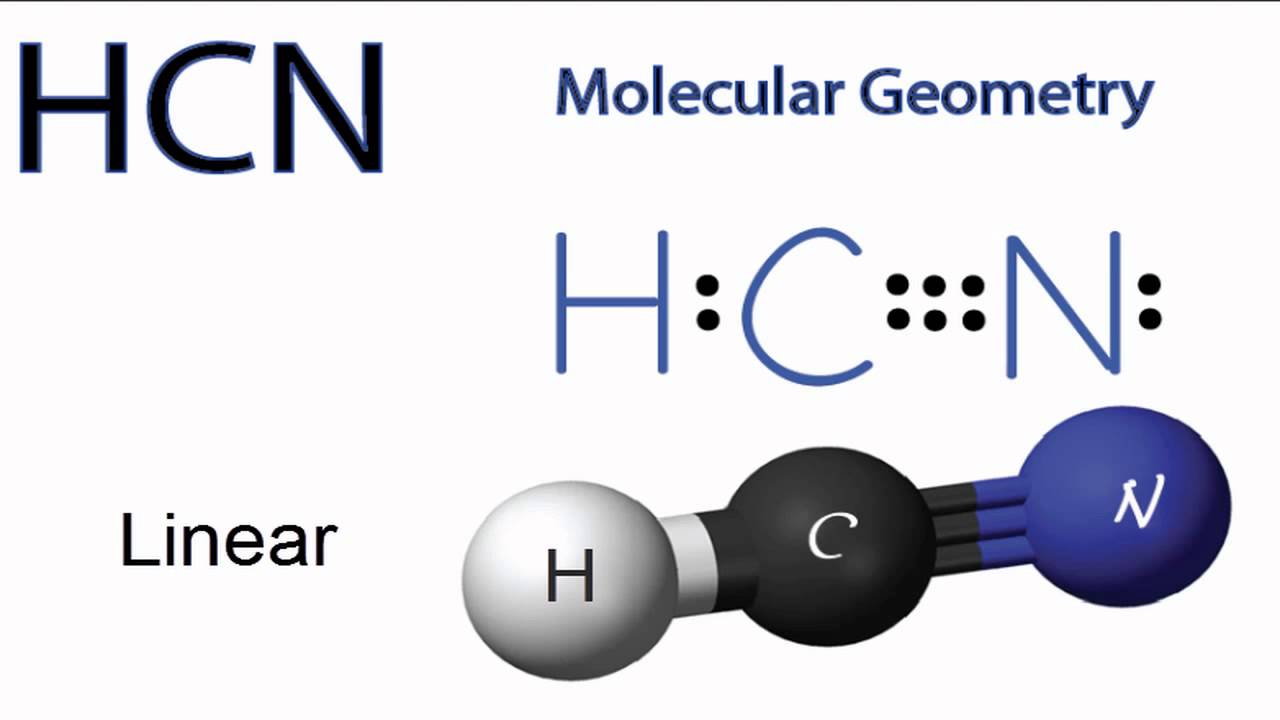
HCN Molecular Geometry YouTube
Molecular Structure of Compounds 6.2 Lewis Structures We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Figure 1 shows the Lewis symbols for the elements of the third period of the periodic table.

HCN Lewis Structure (Hydrogen Cyanide) Molecules, Chemical formula, Lewis
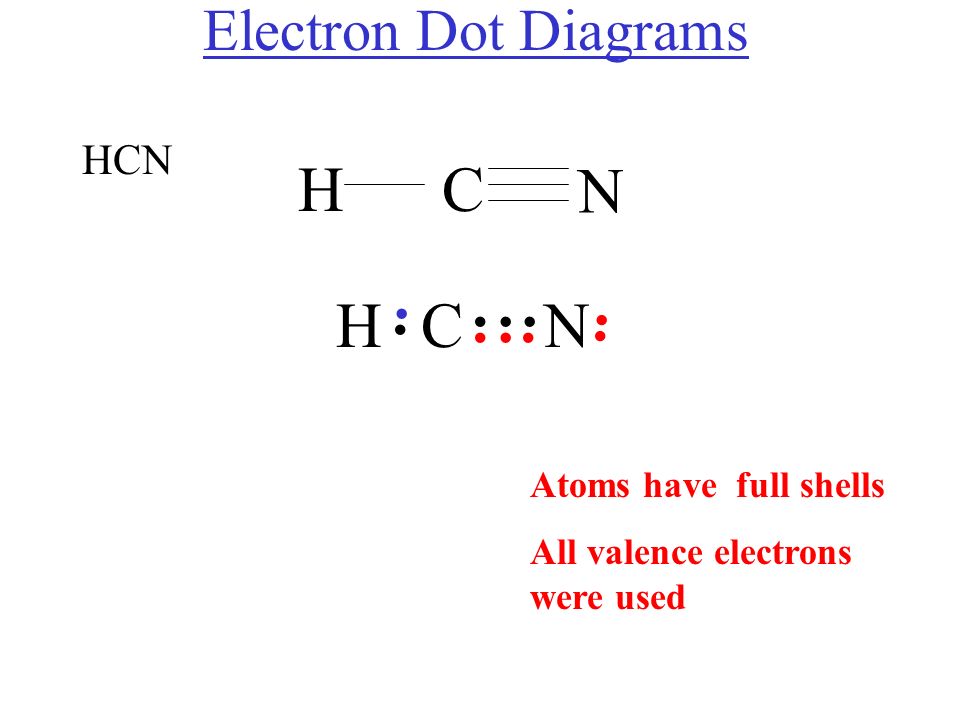
The Lewis dot diagram for HCN is as follows: The hydrogen atom is represented by a single dot, the carbon atom by four dots (arranged horizontally or vertically), and the nitrogen atom by five dots (arranged in a cross shape). These dots represent the valence electrons of each atom. The hydrogen atom shares its electron with carbon, and carbon.
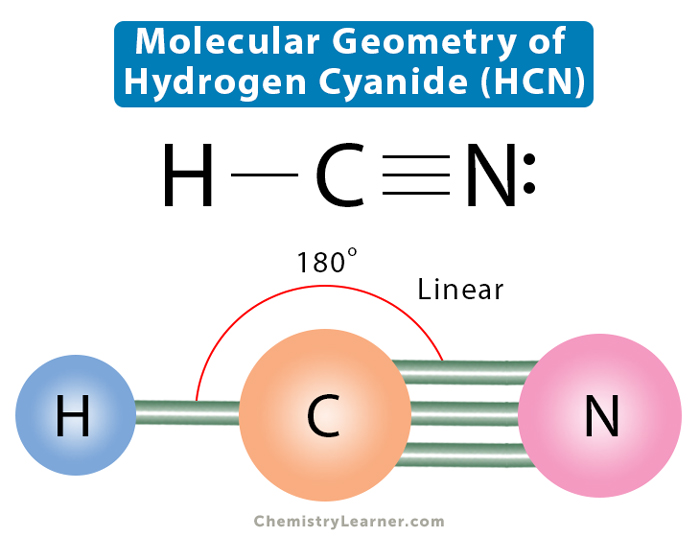
Molecular Geometry, Lewis Structure, and Bond Angle of HCN
The Lewis Structure (Lewis Dot Diagram) for HCN. 1. Count electrons 2. Put least electronegative atom in centre.more.more Lewis Diagrams Made Easy: How to Draw Lewis Dot.
Hcn Lewis Structure Bonds Draw Easy
Use these steps to correctly draw the HCN Lewis structure: #1 First draw a rough sketch #2 Mark lone pairs on the atoms #3 Calculate and mark formal charges on the atoms, if required #4 Convert lone pairs of the atoms, and minimize formal charges #5 Repeat step 4 if needed, until all charges are minimized, to get a stable Lewis structure

[DIAGRAM] Lewis Dot Diagram For Hydrogen Cyanide
The carbon atom has (or shares) 3 electrons from the triple bond, and a lone pair of electrons, which it owns. With 2 inner core electrons, this makes 7 electrons with which it is associated. Since, the atomic number of carbon is 6, the carbon atom is formally negatively charged. When I write H-C-=N:, the carbon atom has a share of 4 electrons.

HCN Lewis Structure, Molecular Geometry, Hybridization, MO Diagram, and Polarity Techiescientist
Transcript: For the HCN Lewis structure we have one valence electron for Hydrogen, we have four for Carbon, and we have five for Nitrogen, for a total of ten valence electrons for the HCN Lewis structure. We'll put the Carbon in the center, because it's less electronegative than the Nitrogen, and Hydrogens always go on the outside of Lewis structures.
Lewis Diagram For Hcn
A quick explanation of the molecular geometry of HCN including a description of the HCN bond angles.Looking at the HCN Lewis structure we can see that there.

lewis dot diagram for hcn Wiring Diagram
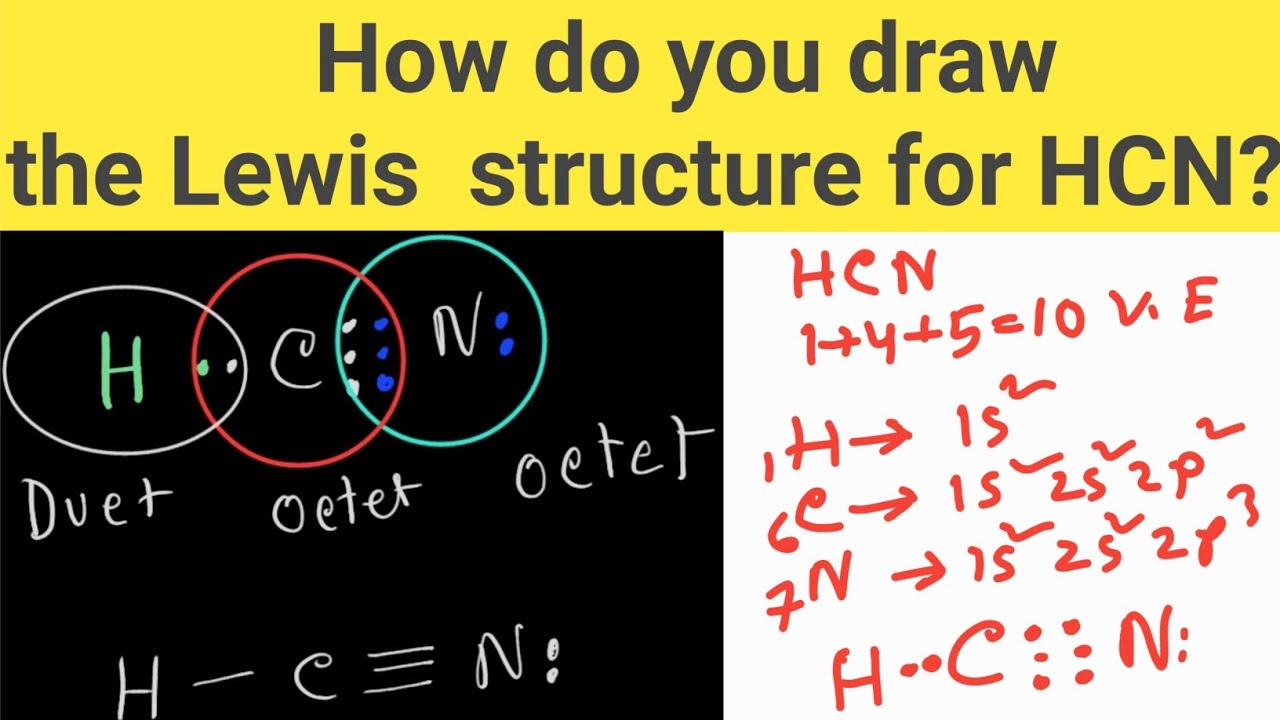
Step #1: Calculate the total number of valence electrons Here, the given molecule is HCN. In order to draw the lewis structure of HCN, first of all you have to find the total number of valence electrons present in the HCN molecule. (Valence electrons are the number of electrons present in the outermost shell of an atom).

Hcn Lewis Structure Bonds Draw Easy
To determine the HCN Lewis Dot Structure first we need to look for valence electrons in individual atoms. Hydrogen (Atomic number = 1 and electronic configuration = 1) belongs to the 1 st group of the periodic table and consists of only 1 electron.

4. 11C04.1 PSV 1 Lewis structure of HCN YouTube
Solution. Steps for Writing Lewis Structures. Example 3.4.1 3.4. 1. 1. Determine the total number of valence electrons in the molecule or ion. Each H atom (group 1) has 1 valence electron, and the O atom (group 16) has 6 valence electrons, for a total of 8 valence electrons. 2.
How do you draw the Lewis structure of HCN (hydrogen cyanide)? HCN Lewis Dot Structure YouTube
Hydrogen Cyanide is a colorless, flammable, and poisonous chemical liquid. Represented by the chemical formula, HCN is one of those molecules that has an interesting Lewis structure. This liquid is used in electroplating, mining, and as a precursor for several compounds. HCN Lewis Structure (Hydrogen Cyanide) Watch on 0:00 / 2:55

Diagrama De Lewis Hcn Estudiar
Steps for Writing Lewis Structures. Calculate the sum of the valence electrons in the molecule. 1 C atom = 1 × 4 = 4 valence e -. 1 O atom = 1 × 6 = 6 valence e -. 2 Cl atoms = 2 × 7 = 14 valence e -. sum of valence e - = 24 valence e -. Construct a skeleton structure for the molecule. C is the central atom since it makes the most.

HCN Lewis Structure How to Draw the Dot Structure II lSCIENCE ll NCERT ll Rohit Sir YouTube
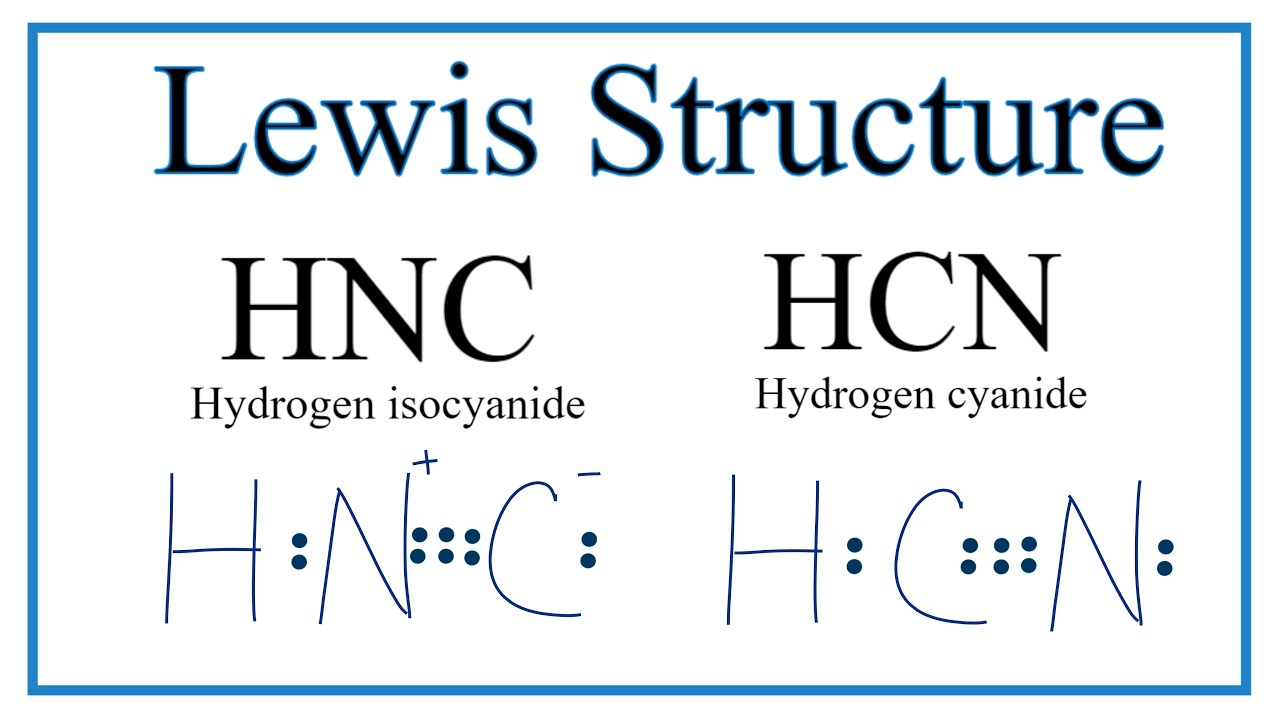
A Lewis structure is a way to show how atoms share electrons when they form a molecule. Lewis structures show all of the valence electrons in an atom or molecule.. Example: calculating the formal charges in HCN and HNC. For the arrangement HCN, the Lewis structure: H-C\(\equiv\)N: